Product D-value Study Part 1: Non-heat Sensitive Aqueous Liquid
By Laurent BERLIET and Silvia RIZZI from Mesa Labs France
[ Read in: Spanish | French | Italian ]
When sterilizing an aqueous liquid product with steam, we generally validate its sterilization by using a liquid submersible commercial Biological Indicator (BI) compliant with ISO 111138-3 suspended in the coldest position of the liquid mass (Spore News Vol-7, No-5), showing whether specified sterilization conditions have been correctly achieved. Biological Indicators such as MagnaAmp or SterilAmp consist of a sealed glass ampoule, with spores of Geobacillus stearothermophilus or Bacillus subtilis 5230 inoculated in a culture medium with a pH color indicator which will change color if spores survive. These BIs have a resistance as described in the standards, measured in a resistometer (ISO 18472) and expressed by the D-value and a population that characterize each manufactured batch.
Depending on heat-sensitivity of the liquid product to be treated, there are two common approaches used, the Overkill method and the Bioburden/Biological Indicator method. Overkill method is the preferred method (the simplest) when items being sterilized can withstand extended exposure to the sterilizing process. It can be validated using a BI compliant with ISO 11138 with a D121-value ≥ 1.5 min and a population ≥ 105 ; however, a 106 population is more commonly used and recommended. This method can inactivate a high concentration of very resistant BI organisms and does not require constant monitoring and control of bioburden.
For heat-sensitive products and with low F0 cycles, the Bioburden/BI method is preferred with the potential of using a liquid submersible biological indicator (BI) inoculated with a lower population (104) of G. stearothermophilus or B. subtilis 5230 (a less resistant organism).
In this Spore News we will discuss the non-heat sensitive products sterilized using Overkill method, while the heat-sensitive products and BI-Bioburden method will be discussed in the next issue. These different validation methods are addressed in ISO 11138-7, United States Pharmacopoeia (USP) and European Pharmacopoeia (EP).
Why would one be interested in performing a Product D-value study for an overkill cycle in which they are using a BI compliant with ISO 11138-3?
As pointed out in the recent versions of USP and EP, properties of the material being sterilized can significantly influence microorganism resistance / lethality especially for liquid products.
USP 40 1229.2 Moist Heat Sterilization of Aqueous Liquids
The selection of a BI must be considered carefully because of the balance that must be maintained between attaining sterilization and maintaining the sterilized material’s essential quality attributes. The biological challenge is either directly inoculated into a liquid-filled container or is introduced via self-contained units provided there is adequate correlation between their resistance and the resistance that would occur in the process fluid. The liquid can be either the product or a surrogate fluid. The resistance of the indicator in the product (and surrogate fluid, where used) must be known. The surrogate’s physical properties should approximate those of the product. If there are surfaces within the container that are not presterilized, biological challenge of those surfaces may be required.
LIQUID D-VALUE DETERMINATION
Determination of the thermal resistance (D-value and z-value) for the biological indicator in the liquid is required. This must be performed in a Biological Indicator Evaluation Resistometer (BIER) in replicate. The thermal resistance of each BI lot in the liquid should be determined. When a surrogate liquid is used for convenience (e.g., a master solution approach) or because of microbial inhibition of the BI by the liquid, the thermal resistance in the surrogate must be determined.”
European Pharmacopoeia 9.2 :2017
5.1.2.2 “Spores inoculated into a product or onto surfaces are known to react differently to sterilizing conditions as compared to biological indicator units. In these cases, commercially available biological indicator units may not be suitable to test sterilization effectiveness and an inoculated test product/item prepared from a well-characterized spore suspension may be a better model to evaluate the effectiveness of the sterilization cycle.”
Since the resistance for a standard steam BI is measured on G. stearothermophilus inoculated into culture medium with pH indicator sealed in an ampoule of variable size, the question that should be addressed would be: what would the D-value be if G. stearothermophilus spores were directly inoculated into the product? Will the D-value of spores in direct contact with the product be lower or higher than the D-value stated on the Certificate of Analysis of a standard BI?
The D121-value for liquid ampoule BIs is generally between 1.5 and 2.5 min. The aim of a D-value analysis on liquid products is therefore evaluating the impact of the product on microorganisms to ensure that the BI used to validate the cycle represents a correct challenge to guarantee an acceptable Sterility Assurance Level (SAL) on the sterilized product. This preliminary study allows one to determine as accurately as possible the cycle to be applied to obtain a Sterility Assurance Level of 10-6 and therefore to be able to claim and guarantee the sterility of its product. It is essential to carry out this study during the development of the product. For existing cycles, a product D-value study will allow one to confirm the choice that has been made historically to comply with current standards.
Let us start by considering a non-heat-sensitive, filterable liquid product, sterilizable with an Overkill approach (e.g., media). A certain number of glass ampoules (or pre-filled devices) containing the product to be analysed are inoculated with a population of 106 G. stearothermophilus spores per ampoule and sealed.
To compare the D-value obtained in the product to the D-value of a standard liquid submersible BI, it is recommended to select a spore crop that has been used to manufacture the standard BIs and to perform the test under the same parameters with the same equipment used for testing these standard BIs. Similarly, when possible, for small volumes (pre-filled syringes, ampoules), the study should be carried out on the complete device including the container. For this reason, it is desirable to carry out Product D-value testing by the manufacturer of the BIs that will be used for validation. If this is not possible, then it is imperative that the third-party laboratory carry out a parallel comparison with the liquid submersible BI under its own conditions to obtain comparable results.
Mesa’s Contract Studies Laboratory has acquired a high level of expertise and experience over the years by specializing in the execution of D-value studies on liquid pharmaceutical products, stoppers, and even complete devices (such as pre-filled syringes) to integrate all the constraints related to sterilization. All our procedures are USP/ISO compliant and carried out in an ISO 13485 certified laboratory, and it is equipped with different test vessels that are compliant with ISO 18472.
Most of these aqueous products have a composition or texture that will not be modified during the cycle, so a linear death kinetic is achieved in the same way as a standard BI (ISO 11138-1 with r2 > 0.8).
When the D-value of a pharmaceutical product is analysed, two types of results are expected:
D-value measured in the product is less than or equal to a standard liquid BI D-value (not significantly different). Then a standard BI represents a worst case, and it can be used for periodic qualification and validation: See Figure 1.
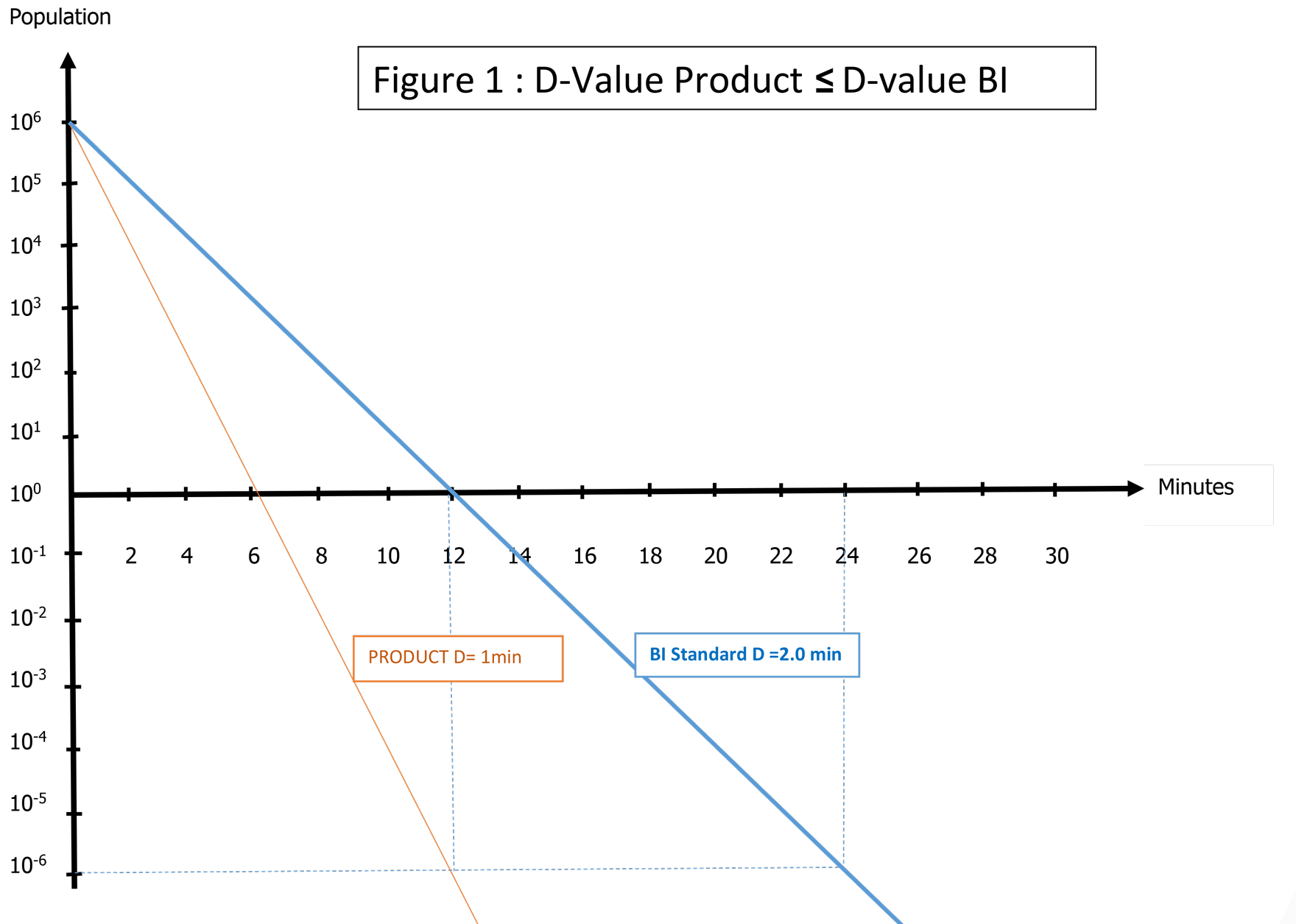
In this example, the G. stearothermophilus inoculated into product has a D121-value of 1 minute; therefore, the use of a BI ampoule with a D-value between 1.5 and 2.5 minutes is an appropriate choice.
D-value measured in the product is higher than a standard liquid BI D-value (> 2.5 or 3) then it will be difficult to find BIs with equivalent or higher resistance. In this case, the BI does not represent a worst case: See Figure 2.
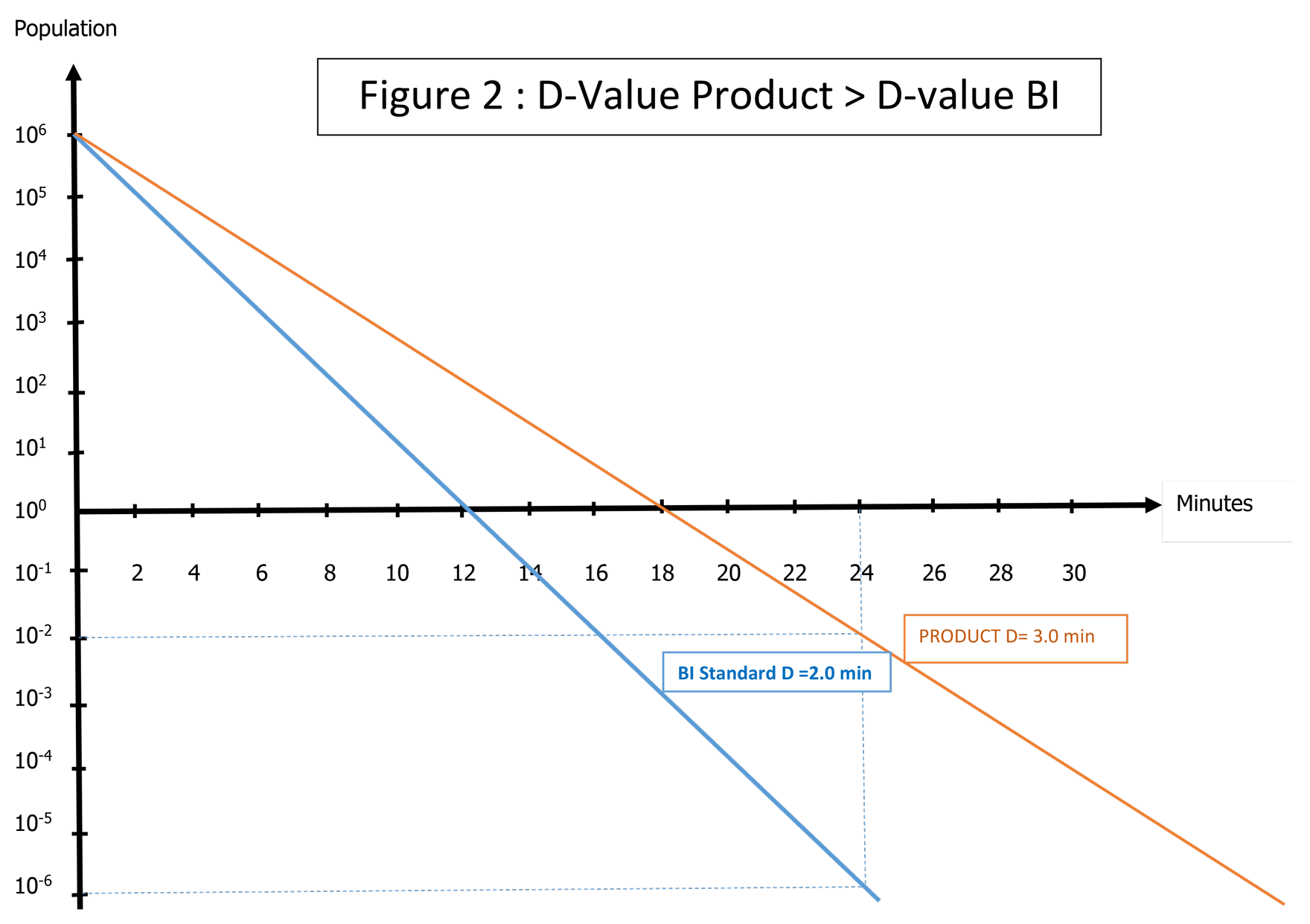
In this example, the appropriate solution is to validate the cycle by direct inoculation of the product with G. stearothermophilus spores. This may generate difficulties in terms of feasibility, homogeneity, certification, spore stability, recovery of spores, etc. The inoculation can be done from a spore suspension with a population of 106 or 107/0.1ml, requiring validation of the inoculation method to guarantee the homogeneity of the inoculum into the product. When validation of the inoculation method is not possible with a spore suspension, it is possible to inoculate spores by the introduction of a naked strip directly into the liquid. This will ensure the intended population is certified.
A third pattern could be considered if bioburden is well monitored.
If the bioburden is lower than 102 CFU for example, then with 8 SLRs an SAL of 10-6 can be claimed. In this case and in theory, a standard BI with G. stearothermophilus 106 can be used to validate an equivalent destruction of 8 SLRs on G. stearothermophilus showing higher resistance due to the product. See Figure 3. This method balances the lower resistance of the standard BI with a larger population than the real bioburden. Of course, the resistance gap must not be too high (<50%) to apply this comparison, which is based on theoretical correlation, and might not be accepted by some auditors or authorities since it goes beyond the framework defined by the validation methods.
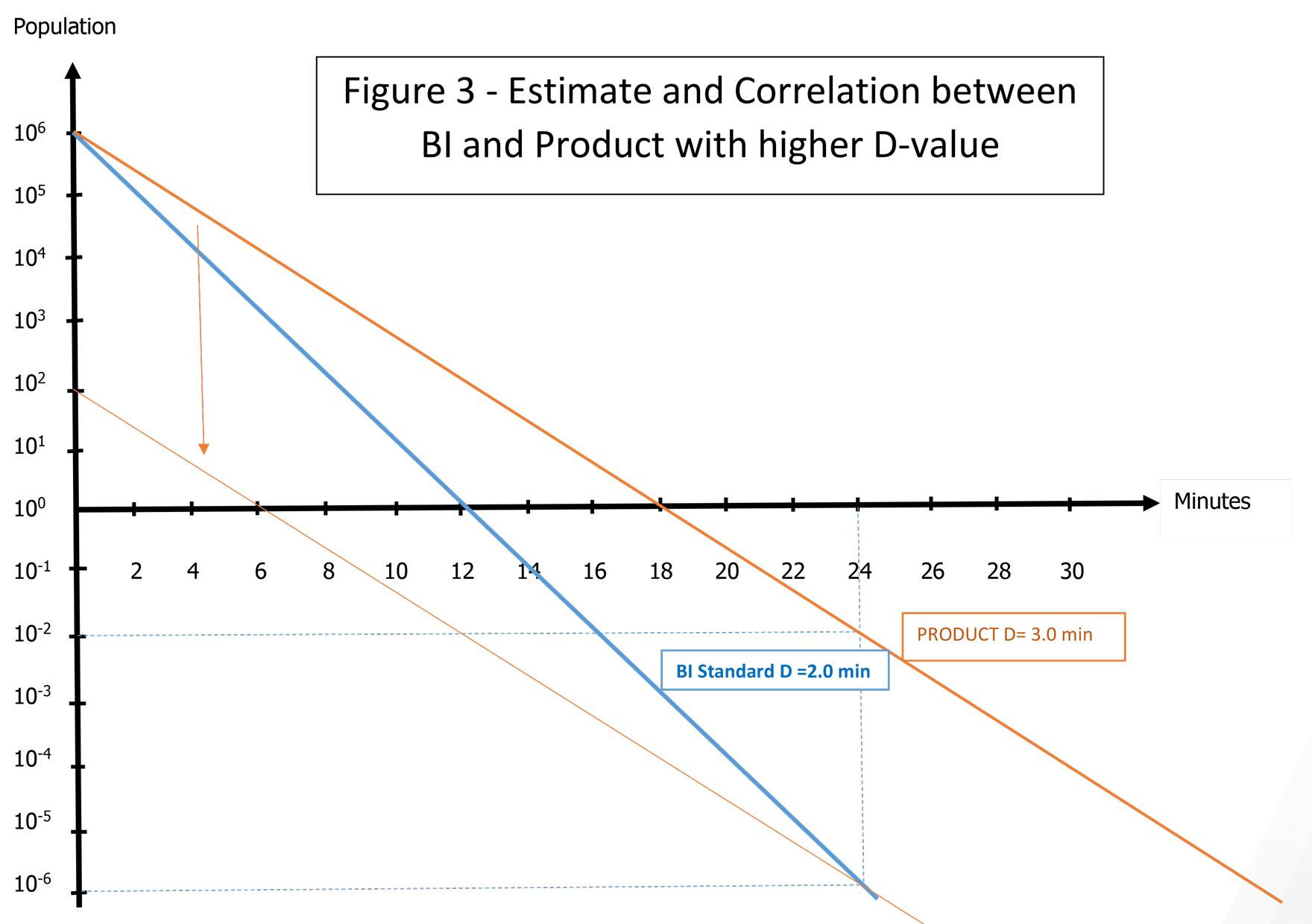
In the next Spore News, we will have the opportunity to discuss other study cases such as heat-sensitive products and atypical aqueous liquids products.