ViewPoint® Continuous Environmental Monitoring Software
Scalable, customizable oversight and reporting
-
Continuous Monitoring Software
Flexible 360° Protection
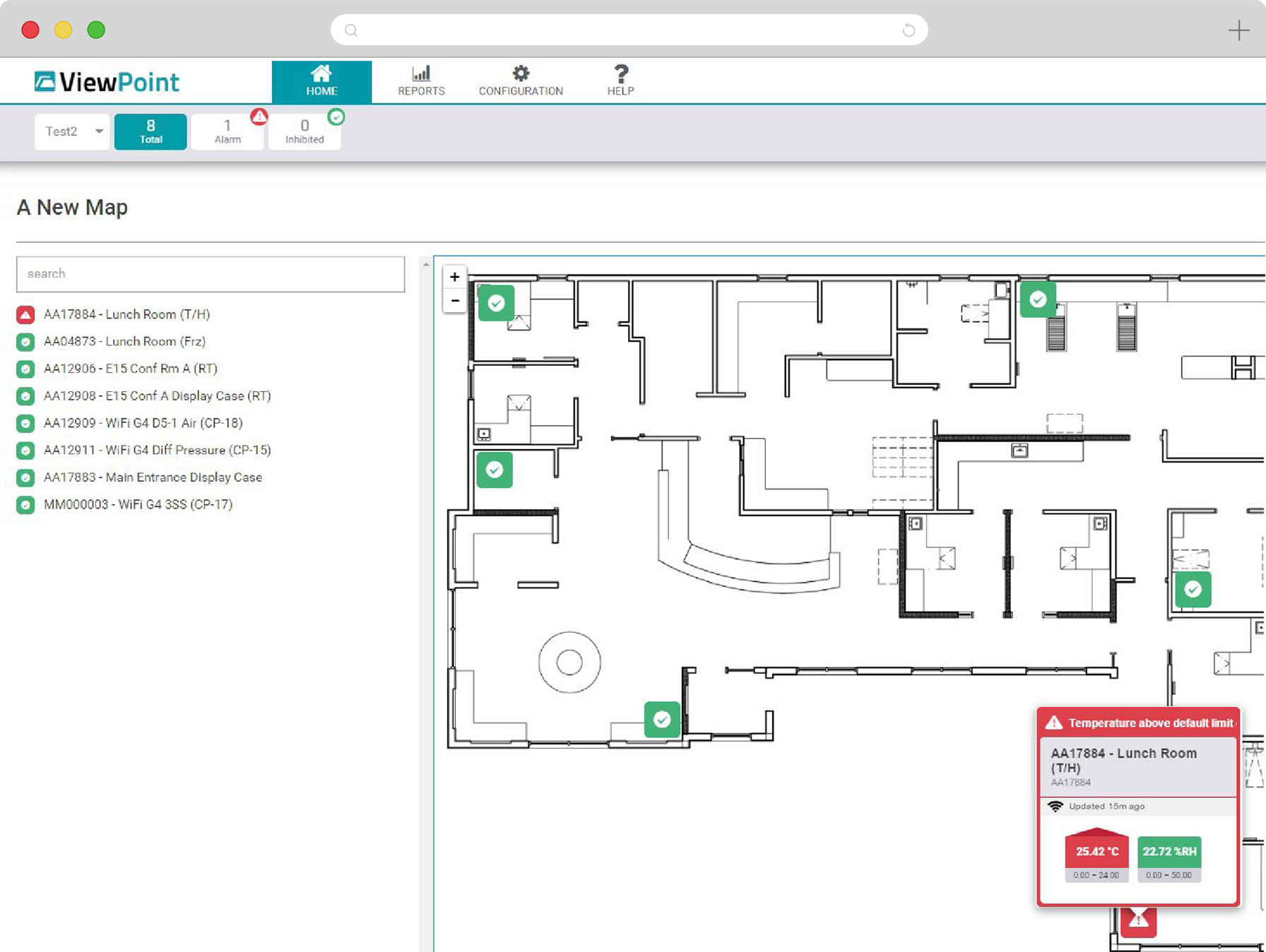
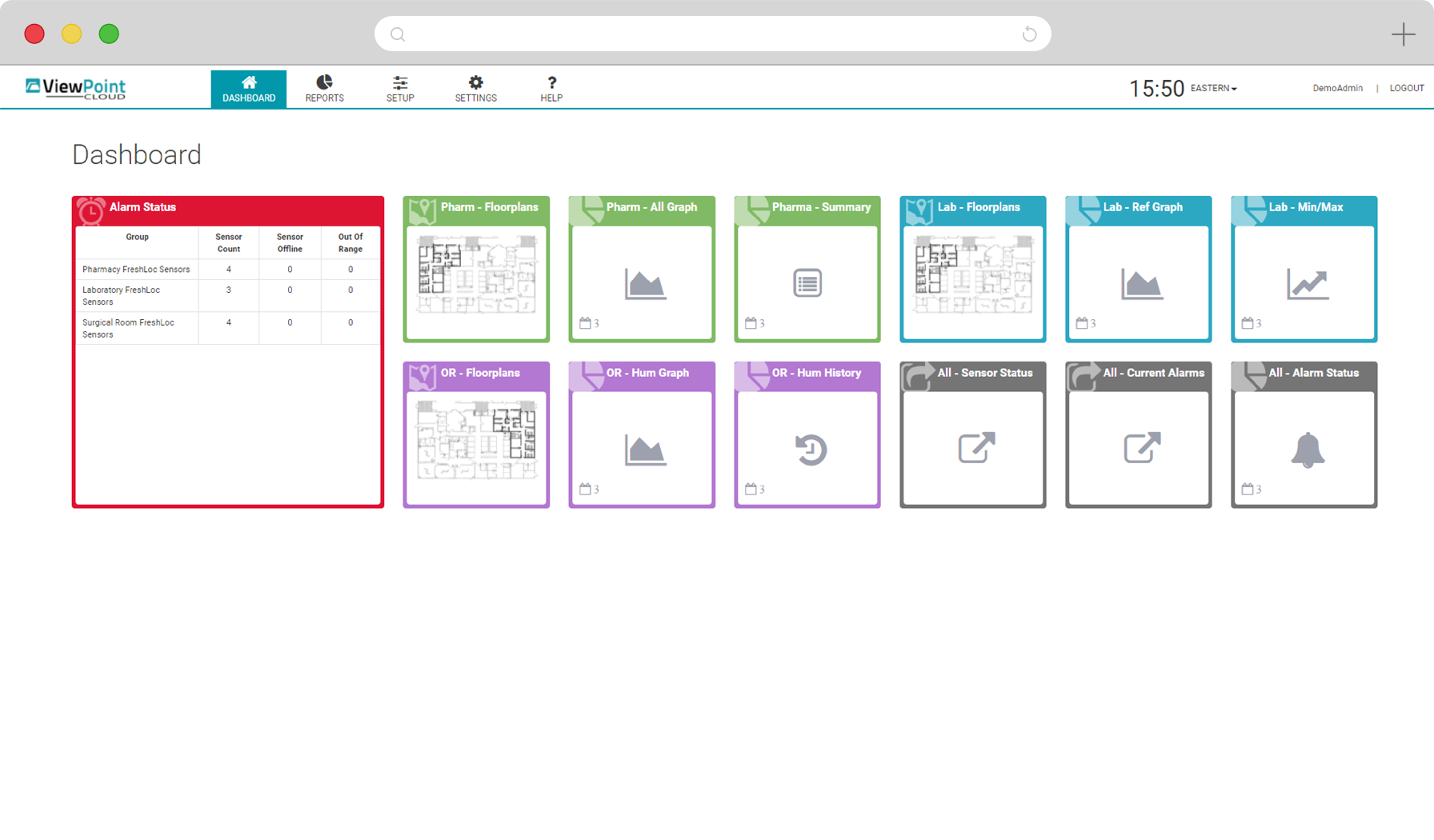
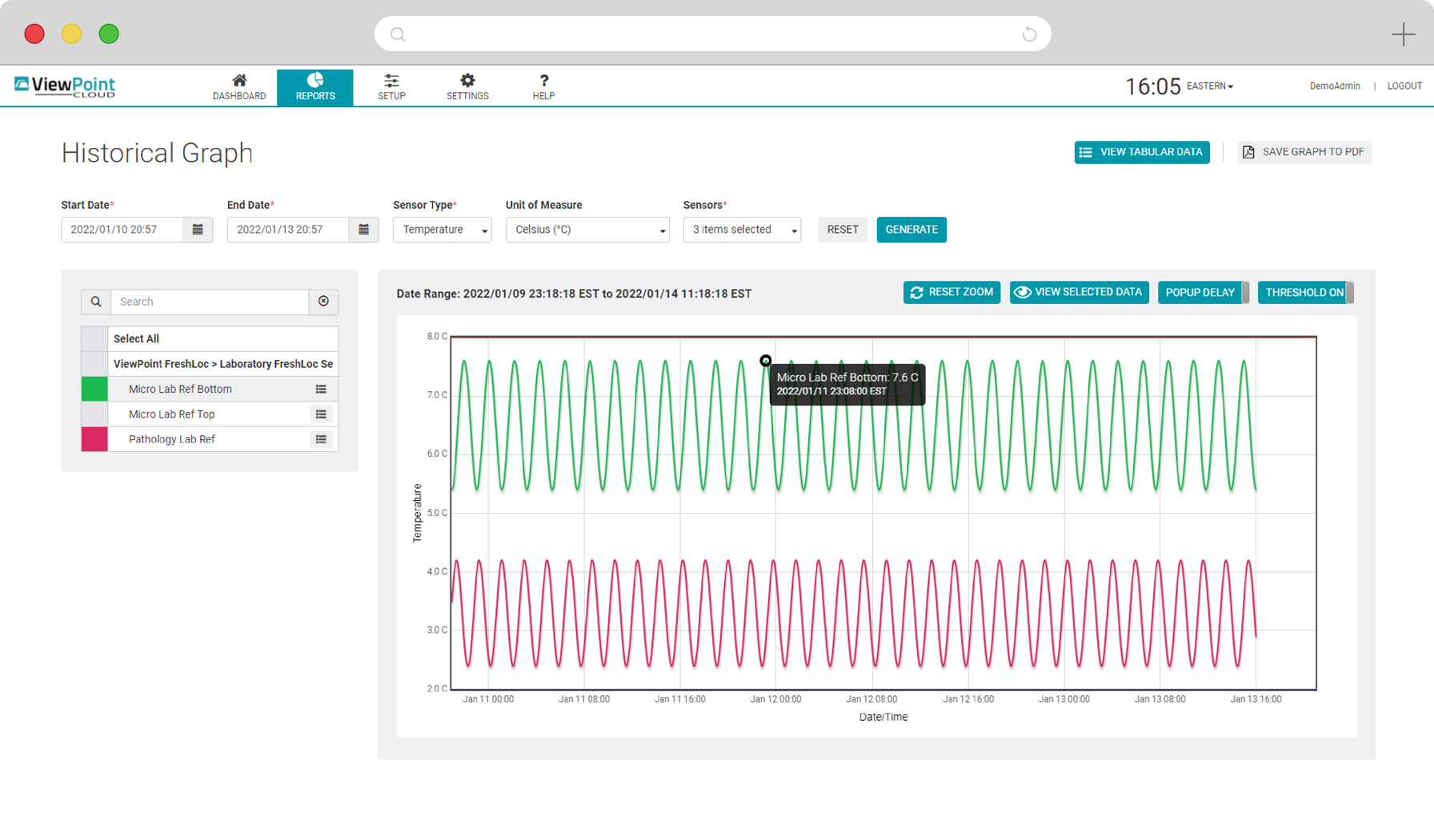
Real-time, gap-free monitoring of multiple parameters and configurable alarming across critical assets, areas and sites, 24 hours a day, 7 days a week. Access live and historical data, reporting and graphs through a single, intuitive, web-based interface.
Scalable, Fit-for-Purpose Solutions
State-of-the-art VPx Sensors and Probes permit real-time monitoring of up to 13 environmental parameters with high degree of accuracy. Choose from cloud-based and on-premise software options to meet your organizational needs and regulatory requirements. Easily add and configure thousands of sensors and manage them all in a single, convenient web application.
Rigorous Compliance
ViewPoint CM Systems conform to regulatory requirements and best practices defined by FDA 21 CFR part 11 and the International Society for Pharmaceutical Engineering (ISPE) published guidelines for Good Automated Manufacturing Practice (GAMP 5). ViewPoint CM System devices are manufactured and calibrated to an NIST1-traceable standard with an ILAC-MRA2 ISO/IEC 17025:2005 certification.
Our software options
Ready to learn more
Contact Mesa Labs to get a custom view of how ViewPoint could work in your context.
Download ViewPoint software, release notes and reference material
Password required, reach out to your Mesa Labs representative for access
Specifications
Compliance
- ISO 17025 calibration laboratory
- FDA 21 CFR Part 11 functionality
- NIST-traceable calibrations
- IQ/OQ/PQ services and documentation
Compatibility
- Current G3 CheckPoint hardware
- Current G4 CheckPoint hardware
Related Products

ViewPoint® Connect
ViewPoint® Connect provides a central connection for a new or expanded hardwired temperature monitoring with enhanced processing and larger memory storage.
Learn more

ViewPoint® VPx Wi-Fi Sensor
Available with LCD display and wireless options, VPx offers more capacity, convenience, and security in a VFC-compliant temperature monitoring solution.
Learn more