
How to Prove a Sterility Assurance Level of 10⁻⁶ PART 2: The Biological Indicator/Bioburden Method

Written by Kurt McCauley and Laurent Berliet
Sterile product is defined as material (i.e., final product, product components, etc.) that is free of viable microorganisms. There are numerous sterilization modalities in use and several recognized approaches for achieving a state of sterility, the most popular of which is the overkill half-cycle method (Spore News - How to Prove a Sterility Assurance Level of 10⁻⁶ PART 1: The OVERKILL Method(s)). However, another approach, the biological indicator/bioburden (BI/BB) method is gaining in popularity primarily due to the increased development of heat-sensitive products, and/or the need to validate sterilization processes (e.g., ethylene oxide) that use reduced amounts of sterilant (for employee and environmental safety reasons).
Note: A third bioburden-based method exists and is briefly described later in this paper.
The BI/BB method is based on the resistance characteristics and population of the product bioburden as opposed to the hyper-resistant characteristic of a theoretical bioburden (as is the case with the overkill method). As the name implies, the overkill method delivers lethality to the load well in excess of that actually needed to inactivate the bioburden, while the BI/BB approach is designed to deliver sufficient lethality to render the product sterile without excess insult to the product.
The BI/BB method requires extensive knowledge of the product bioburden whereas the overkill method does not. Bioburden characteristics such as numbers, types, and resistance should be well understood, as the sterility assurance level (SAL) of the validated sterilization process will be based on these attributes. Additionally, stricter control of components and assembled product prior to sterilization will minimize the level of bioburden which allows for a reduced process time while still demonstrating the appropriate SAL. However, regarding moist heat processes, the European Pharmacopoeia section 5.1.1, Methods of preparation of sterile products, states: “The minimum F₀, calculated in the slowest-to-heat position of the load is not less than 8 min.”
Cycle development activities are based on the bioburden characteristics and the influence the product has on these organisms (e.g., product D-value studies), while the validation of the process utilizes biological indicators to demonstrate the SAL. The surrogate test organism used in these BIs is typically Bacillus subtilis 5230 (moist heat) which is less resistant than the strain used for the overkill method (Geobacillus stearothermophilus). More resistant strains can still be used at either a reduced spore count or by direct enumeration at the targeted exposure. This is supported by USP <1229> which states "an approach in which the incomplete destruction (or destruction of a modest population) of a resistant BI can be used to demonstrate the capability of the method to reliably destroy the bioburden present." Figure 1 compares the resistance of a bioburden isolate and three configurations of biological indicators.
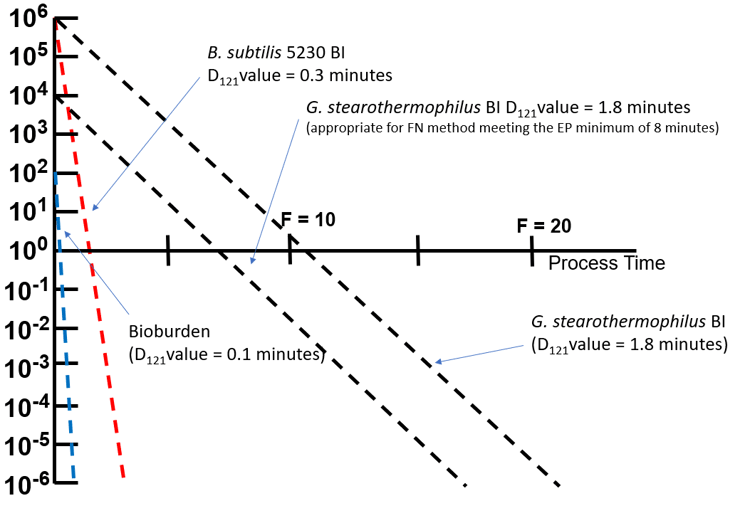
Figure 1. Moist heat resistance comparison of a bioburden isolate and three types of biological indicators.
Several standards give guidance on the BI/BB method including:
-
ISO 17665 Sterilization of health care products—Moist heat—Requirements for the development, validation and routine control of a sterilization process for medical devices
-
ISO 11135 Sterilization of health care products—Ethylene Oxide—Requirements for the development, validation and routine control of a sterilization process for medical devices
-
ISO 20857 Sterilization of health care products—Dry heat—Requirements for the development, validation and routine control of a sterilization process for medical devices
-
ISO 14937 Sterilization of health care products—General requirements for characterization of a sterilizing agent and the development, validation, and routine control of a sterilization process for medical devices
-
ISO 11138-7 Sterilization of health care products—Biological Indicators—Part 7: Guidance for the selection, use and interpretation of results
-
United States Pharmacopeia <1229> Sterilization of Compendial Articles
Several prerequisite studies are needed in order to develop the sterilization process including bioburden characterization (population, identification, and boil test to screen for spore formers), product D-value studies, and mapping the load to identify areas of lower lethality.
Bioburden characterization
The types and numbers of microorganisms present should be determined on successive lots of product, and continuously monitored over time as bioburden characteristics may change with the seasons. The bioburden can contain mesophiles, thermophiles, aerobic and anaerobic organisms, and appropriate recovery methods should be employed during the screening process. ISO 11737 Determination of a population of microorganisms on products, provides guidance on these activities. Of particular interest will be the presence of spore formers which are isolated from the bioburden via a boil test. It is the spore formers that will be most resistant to the sterilization process and should be the focus of further characterization. Vegetative organisms are significantly less resistant to environmental stresses and are of lesser concern unless they are present in significantly high numbers (which indicates poor control of a material(s) and/or environment). A common practice when recovering spores is to identify the most resistant of the organism(s) to the genus or species level. These organisms are then evaluated for their resistance in the product D-value studies.
Product D-value Studies
Product D-value studies evaluate the effect the product and/or closure components have on the resistance of the microorganisms. Additional information on Product D-value studies can be found in (Spore News – Why Perform a Product D-value Study? Part 1) and Spore News - Product D-Value Study Part 2 - Heat-sensitive & Atypical Aqueous Liquids)
Load Mapping
Load Mapping identifies the areas of lower lethality in a worst-case validation load. For thermal processes, this is typically accomplished by thermal mapping of the load during a process to identify the cold spots. For chemical sterilization processes, additional data on critical parameters is necessary and may include the use of RH and pressure data loggers as well as chemical indicators. The continuous control of these parameters during the cycle is required to guarantee the effectiveness of the sterilization process. The physical validation of the process ideally would demonstrate the homogeneity of conditions at all points within the load. Complete uniformity within the load is not possible and it is important to identify the areas of lower lethality which are then the focus of the biological testing.
Cycle development and validation
Once the above-mentioned studies are complete, the cycle development and validation process can begin. The cycle development activities include calculating the exposure time required to achieve the targeted SAL utilizing the bioburden population and D-value. The cycle validation activities demonstrate that the necessary lethality is delivered to the targeted locations during the exposure time using biological indicators. The output from the biological indicators can either be in the form of direct counts of surviving organisms or most probable number (MPN) testing using replicate BIs at each test location (Figure 2.)
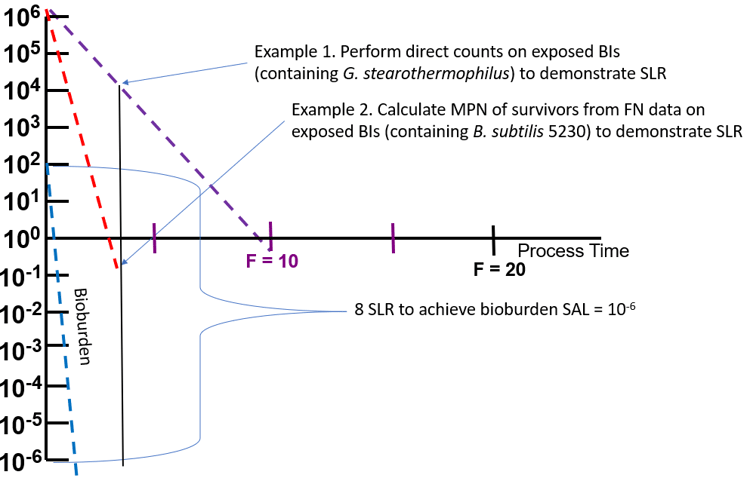
Figure 2. Example of bioburden SAL and two biological indicators options for confirming lethality
The following is an example of the mathematical process:
1. Bioburden characterization and isolation of spore former from boil test
-
Identification: (B. amungus) spore former
-
Population (N0) = 1 x 102
-
D-value (D) in product = 0.1 minutes but rounded up to 0.3 as a safety factor
2. Calculate bioburden exposure (F) to achieve desired SAL (e.g.; 10-6) using bioburden population (N0), D-value (D), and Spore Log Reduction (SLR)
-
F = SLR X D
-
Calculate SLR using formula: SLR = log N0 – log (SAL)
-
SLR = log 1.0 X 102 - (-6) = 8
-
-
Exposure time to achieve SLR of 8 is F = SLR X D
-
F= 8 X 0.3 = 2.4 minutes
-
3. Obtain BI with appropriate characteristics, for example:
-
Identification: (B. subtilis 5230)
-
Population (N0) = 2.0 x 106
-
D-value (D) = 0.4 minutes
4. Place BIs in areas of lower lethality as determined in the load mapping experiments, for example:
-
Three BIs in Location 1
-
Three BIs in Location 2
-
Three BIs in Location 3
Note: BI performance after treatment can be evaluated by either direct count or MPN calculations. This example uses the MPN method.
5. Perform exposure (F) greater than that needed for bioburden SAL (10-6), for example:
a. Bioburden exposure (F) to achieve a SAL of 10-6= 2.4 minutes
b. Run 3-minute exposure
c. BI result after suitable incubation period:
-
-
Location 1-->3 growth negative BIs/3 replicate BI
-
Location 2-->1 growth negative BIs/3 replicate BIs
-
Location 3-->2 growth negative BIs/3 replicate BIs
-
d. Location 2 provides the greatest challenge to the process and the MPN of surviving spores from that location is used in the calculation:
MPN = ln (n/r) where
n = number of replicate BIs
r = number of growth negative BIs
MPN = ln (3/1)
MPN = 1.1 spores per unit
6. Calculate SLR of the process based on the BI as follows:
a. SLR = log N0 – log Nf = log 2.0 x 106 – log 1.1 = 6.26 This demonstrates an ~ 6SLR.
7. Qualification cycle
- Increase the exposure time to 4 minutes to achieve all growth negative BI
- Run 3 replicate 4-minute exposures to demonstrate reproducibility
8. Calculate SAL of bioburden
- SAL = 10(log No -SLR) where N0 is the bioburden population
- The bioburden SLR = F/D, = 4.0/0.3 = 13.3
- SAL = 10 (log No -SLR) = 10log 100 – 13.3 = 10-11.3
Figure 3 provides a graphical representation of this example.
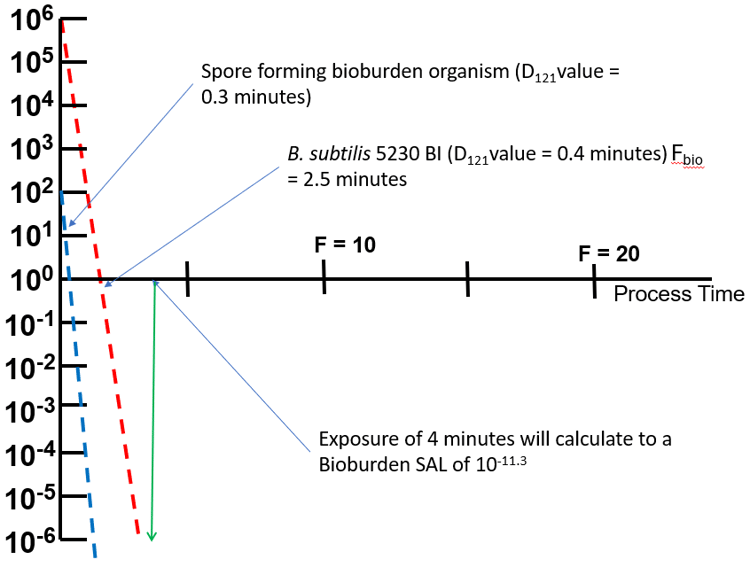
Figure 3. Graphical representation of mathematical example
In summary, the BI/BB method of validation requires additional precursor studies as compared to the overkill methods; however, once these are complete, the ability to develop a cycle that truly targets the contaminating organisms can be achieved. This is especially important when the product is sensitive to the sterilization process and is damaged by excessive treatment that is inherent with the overkill methods. Alternately, the method can be used to validate sterilization processes (e.g., ethylene oxide) that use reduced amounts of sterilant for safety concerns. The BI/BB method requires greater knowledge, stricter controls, and ongoing monitoring of the bioburden while still allowing the ability to establish a SAL of 10-6.
Bioburden-based method
The bioburden method is based on the inactivation of the naturally occurring organisms during defined incremental exposures to the sterilization process. The method requires detailed knowledge and control of the bioburden (as described with the BI/BB method) including characterizing the most robust of the bioburden organisms. The method does not develop a process based on a sterility assurance level and has other limitations compared to the BI/BB and overkill methods.
In brief, a process is developed by treating product to incremental conditions that result in both non-sterile product (at shorter exposures) and sterile product (at longer exposures). Product is tested for sterility per the methods in ISO 11137 Determination of a population of microorganism on products and per a defined sampling plan (number of units tested/number of units exposed). It is here where the limitations of the process occur as the conditions for microorganism recovery are numerous (mesophilic, thermophilic, aerobic, anaerobic, etc.) and the percentage of units sampled would need to be large to increase the detectability of non-sterile product. Additionally, sterility testing is both time-consuming and destroys the product under test.
Mesa Labs provides a wide range of biological indicators for use in process development and validation including custom BIs that can be used for monitoring the areas of lower lethality in product loads. Additionally, the Contract Studies lab at Mesa routinely performs bioburden characterization and product D-value studies, both of which are key components when validating a process by the biological indicator/bioburden method.
SN081-V1