Smart-Read® EZTest® Biological Indicators
-
Smart-Read EZTest Biological Indicator
Smart-Read® EZTest®: Fast and Reliable Sterile Product Release
A fast and reliable solution to confirm sterility of products. With no added enzymes or chemicals, this unique system uses a real biological indicator (BI) in a simple, automated process. Detect sterilization failure in just 3-5 hours, with negative results confirmed in 10 hours. You can expect:
- Fast biological results
- True biological system
- Easy one-step evaluation
- Automatic documentation
- Alarm on sterilization failure
- 100% verifiable results
The Smart-Read system includes the Smart-Read EZTest self-contained biological indicator and Smart-Well® incubator for rapid evaluation. Easy to implement with minimal training, this system ensures fast, accurate, and verifiable results, with automatic documentation and alarms for sterilization failure.

Smart-Well® Incubator
The heart of the Smart-Read Monitoring System, the 1710 Smart-Well is a highly sophisticated monitoring tool that is intuitive and designed with the user in mind. The Smart-Well incubator is easy-to-configure with its simple touch-screen interface and color LCD display. The incubator can evaluate up to ten Smart-Read EZTest biological indicators independently, and contains an additional cell for a positive control unit. Each BI test result is automatically documented with a user-customizable printed report, and an alarm is sounded the moment that sterilization failure is detected.
The 1710 Model Smart-Well offers these enhanced features:
- Dual Integrated Digital Thermometers
- Improved Processor and Storage
- Larger, Color LCD Interface
- Password Protection
- Temperature Progress Meter
The Smart-Well incubator is offered in a kit including the unit, user's manual, report printer, and all necessary cabling.
The Smart-Well® incubator is protected U.S. Patents 6,340,590 and 6,455,272. Other patents pending.

Smart-Read EZTest
The Smart-Read EZTest is a familiar self-contained color change biological indicator specifically designed for rapid evaluation in the Smart-Well incubator. It requires no special processing and can be used in place of any existing self-contained BI. The Smart-Read BI contains spores of Geobacillus stearothermophilus and is suitable for steam sterilization processes.
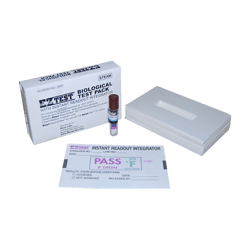
Smart-Read EZTest Test Pack
Smart-Read EZTest Biological Test Pack with Instant Readout Integrator is designed specifically for biological testing of 132°C (270°F) pre-vacuum steam sterilizers. The test pack consists of a self-contained biological indicator containing Geobacillus stearothermophilus inside a small package of porous and nonporous materials, simulating the biological indicator 16 towel test packs defined by ANSI/AAMI ST79. When used as directed, the Chemical Integrator Record Card gives visible indication that sterilizing conditions were met.
“Biological Indicators should be used within PCDs for routine sterilizer efficacy monitoring at least weekly, but preferably daily every day that the sterilizer is in use.”
– ANSI/AAMI ST79
Technical Specifications
Smart-Read EZTest Biological Indicator
Storage
Room temperature
Expiration
24 months from date of manufacture
Certification
Smart-Read EZTest biological indicators are traceable to a recognized culture collection and certified for population, D-value, z-value and performance.
Smart-Read EZTest
| Spore | G. stearothermophilus | |
| Incubation | 10 Hours at 58-62°C | |
| Population | 105 | 106 |
| Units |
100 |
100 |
| Reorder# |
SEZS/5 |
SEZS/6 |
Smart-Read EZTest Biological Indicator Test Pack (PCD) – 25 units per case
| Spore |
G. stearothermophilus |
|
| Incubation | 10 Hours at 58-62°C | |
| Dimensions | 4-1/2″ x 2-5/8″ x 3/4″ | |
| Population |
105 |
|
| Reorder# |
SRSP/05 (5 controls) |
SRSP/25 (25 controls) |
Accessory Specifications
Smart-Well Incubator
Reorder #
US: SWI1710-1
European: SWI1710-3
Australian: SWI1710-4
UK: SWI1710-5
Related products
Related services

Contract studies laboratory
Mesa’s independent contract studies laboratory performs certified and compliant third-party biological indicators certificate of analysis verification.
Learn more

Sterilization cycle development
Get expert help for your bioburden. Mesa Labs assists with sterilization cycle development, BI qualification and validation, and routine monitoring support.
Learn more