Mesa UniQ® Biological Indicators for Industrial Use
-
Mesa UniQ Biological Indicators for Industrial Use
Elevate Sterilization Monitoring in Industrial Spaces
Introducing the UniQ® industrial biological indicators, your ultimate solution for precise sterilization monitoring in industrial spaces, even in challenging situations. While spore strips and ampoules may not be suitable for certain manufacturing scenarios due to size, packaging, or carrier material limitations, Mesa understands the unique requirements of the medical device and pharmaceutical industries. That's why we offer a full line of industrial use biological indicators specifically designed to address these challenges and assist in validating the sterilization process.
- Specialized Solutions: UniQ industrial biological indicators are modified or specially created to cater to the unique needs of the medical device and pharmaceutical industries.
- Flexible and Versatile: Designed to address limitations posed by size, packaging, or carrier material, these indicators provide a reliable solution for monitoring sterilization processes.
- Empowering Validation: Manufacturers and validation specialists can trust Mesa UniQ industrial biological indicators to ensure the effectiveness of sterilization in their specific settings.
Invest in the UniQ industrial biological indicators today and experience precision, efficiency, and peace of mind in your industrial sterilization processes, no matter the size or packaging constraints you may encounter.
Please note that the user must assess the suitability of the indicator for their specific use, as we prioritize empowering you with the right tools for your unique requirements.

Paper Discs
Paper discs (6mm) are used during validation where placement of a strip is impossible due to its size. Suitable for Steam, EO, and Dry Heat.

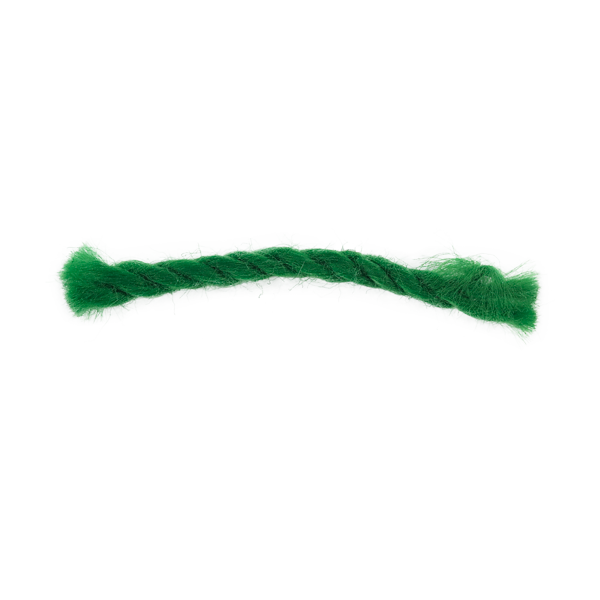
UniQ Thread
Threads (Length: 19mm Diameter: 1.5mm) have been used at the interface of a syringe and plunger to show sterilant penetration and efficacy at that point. Other uses include placement in tubing and small vials. UniQ threads are 100% cotton, 3/4″ long, and are inoculated with B. atrophaeus. The device/lumen into which the thread is inserted affects its resistance characteristics.

UniQ Proline®
Biological Indicator system designed for use in the validation or monitoring of steam sterilization cycles of tubing ranging from 1/8″ to 5/8″ tubing ID. Contains a filter paper disc inoculated with bacterial spores of Geobacillus stearothermophilus.

UniQ MicroStrips
At 2x10mm or 1x25mm, MicroStrips can be placed in small areas where other standard BIs are too large. UniQ MicroStrips are available inoculated with Bacillus atrophaeus, Geobacillus stearothermophilus, or Bacillus subtilis 5230.

UniQ Disc
The stainless-steel Disc (7mm) is inoculated with 103 or 106 B. pumilus spores and packaged in clear plastic. The clear plastic package will not impede UV light and provides protection from post-process contamination.
UniQ Spore Suspensions
Spore suspensions are calibrated suspensions of bacterial spores appropriate for direct inoculation of products or preparing custom biological indicators for monitoring Steam, Ethylene Oxide (EtO), Dry Heat, Hydrogen Peroxide, Radiation and other industrial sterilization processes. Each vial contains 10 ml of suspension. The suspending liquid is 20%, 29%, or 40% ethanol or purified water.
Document Downloads
- Brochure – Mesa UniQ Biological Indicators for Industrial Use
- Product Specifications – Custom MesaStrip Ethylene Oxide
- Product Specifications – Custom MesaStrip Low-Temperature Steam
- Product Specifications – Custom MesaStrip Steam
- Product Specifications – MesaDisc – Steam
- Product Specifications – MesaDisc Radiation
- Product Specifications – SporeDisc – Ethylene Oxide
- Product Specifications – ProLine
- Product Specifications – Cotton Threads – Ethylene Oxide
- Population Assay Instructions – EZTest H2O2 and Stainless Steel Discs
- Population Assay Instructions – Paper, Quartz, & Cotton Thread Carrier Products
- Safety Data Sheet – Mesa Disc and Spore Disc
- Safety Data Sheet – ProLine
- Safety Data Sheet – Cotton Thread
- Safety Data Sheet – Stainless Steel Carrier
- Safety Data Sheet – Stainless Steel Disc
Related products
Related services

Contract studies laboratory
Mesa’s independent contract studies laboratory performs certified and compliant third-party biological indicators certificate of analysis verification.
Learn more

Sterilization cycle development
Get expert help for your bioburden. Mesa Labs assists with sterilization cycle development, BI qualification and validation, and routine monitoring support.
Learn more