VOL 14, ISSUE 2
The MeCo Solution: Improving Consistency, Accuracy and Robustness in SIP Validation

By Laurent Berliet and Silvia Rizzi
USP 1229.13 STERILIZATION-IN-PLACE: “The focus of the SIP procedure is sterilization of the product contact surfaces (the interior of the system). Demonstration of process lethality relies upon physical measurements and biological indicators.This confirmation should extend to the “sterile boundary” of the system, including vessel headspace, connections to other vessels/equipment, and other parts of the system. The interior surfaces of the process equipment, irrespective of their materials of construction, should be exposed to lethal conditions sufficient to sterilize the system and confirmed as lethal with an appropriate biological challenge.”
ISO 13408-5:2006 Aseptic processing of health care products —STERILIZATION IN PLACE: 8.5.1.6 “Biological indicators or inoculated carriers shall be used during the performance qualification. They shall comply with ISO 11138-1 and any other appropriate part of ISO 11138, as applicable (see ISO 14937). The number and locations of biological indicators shall be specified. Documented evidence shall be provided to show that the number and locations of biological indicators are sufficient to demonstrate that the requirements for SIP of the equipment have been met at locations presenting the greatest sterilization challenge.”
Sterilization in place (SIP) is one of the most challenging sterilization processes to control and validate from both a biological and parametric aspect due to complex piping installations, varied steam flow, frequent condensates, and extreme difficulty in placing and controlling the position of the biological indicators (BIs) and thermocouples (TCs), especially in the most difficult to sterilize location. This placement remains complicated, whether accomplished with existing gasket devices or “home-made” systems. The lack of secure placement of the BIs and TCs has resulted in non-conformities caused by inconsistent temperature readings, positive BIs or even shredding and loss of BIs in the system. These non-conformities lead to investigations and potential re-qualifications which increase investment of time and money.
What are the risks related to non-compliant positioning for BIs and TCs during SIP validation?
Unreliable Results
The reproducibility of the TC and BI placement should be completely under control to guarantee comparison of historical validation data. Thermocouples and biological indicators are extremely sensitive measuring instruments. A slight change in positioning could cause deviating test results. If TCs and BIs are not secure in the pipes, they can also come into contact with the walls and condensates which affect the temperature readings and can cause false positive BIs. The current placement methods for TCs and BIs often influence process flows in such ways that test results do not provide an accurate representation of the system operation.
If tape is wrapped around the BI, not only can the tape block the steam from contacting the spores but the removal of the spore strip from the glassine envelope for cultivation will also become very difficult increasing the risk of post process contamination.
Any of these effects can result in inaccurate and unreliable results which trigger deviations and non-conformances.
Damage to Equipment
Due to the lack of quality positioning tools, TCs and BIs are often positioned inside the process system using “do it yourself” materials. Wire mesh, metal wire, traps, and all kinds of uncertified materials are used for positioning during validation studies, causing damage to the sterile, electropolished surfaces of the process equipment. Thermocouples can also be damaged from the stress caused by placement materials.
Contamination of the System
When BIs are placed in direct contact with the surface of the equipment (e.g. surface of vessel, piping, etc.), heat and moisture can cause the ink, used for the text printed on spore strips, to be transferred to the surface of the process equipment causing contamination by ink residues. If tape is used to attach the TC and BI to the predefined test locations, tape remnants and/or adhesive residue can be left behind, causing pollution of the system.
It is a frequent occurrence that spore strips are shredded or even lost during validation cycles due to moisture and high flow rates inside the process system. When this happens, extremely small fibers are spread throughout the system or the BI can get lodged in piping/valves. Not only is there loss of reliable results in these situations, but there is contamination of the entire system and, potentially, the final sterile product.
In order to address all the above issues and non-conformities, Mesa Labs decided to partner with Tyglar company to develop and offer the MeCo solutions for ideal placement of TC and BI during SIP qualifications and validations of aseptic filling processes.
Any contamination of the system will require additional downtime, cleaning and sterilizing resulting in increased cost of validations.
How do MeCo solutions facilitate consistency and accuracy during SIP qualifications and validations?

MeCo Round solution for piping is composed of a hollow round body in a variety of sizes, with caps at both ends. It can accommodate a Mesa BI Spore Strip packaged in a glassine pouch, together with the TC securely fixed by a screw cap. Reproducibility of the TC and BI placement to guarantee accurate data comparison and consistency during validation can be successfully achieved with the aid of a pusher which is attached to the end of the round body. Once placement is determined for each testing location the numbered sections on the pusher allow easy replication of the exact location as described in the test protocol.
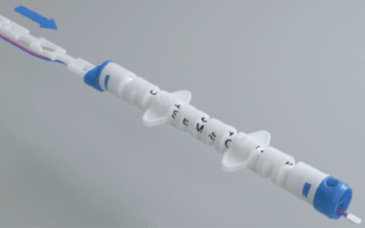
In addition, a system of lateral wings adjusted to the diameter of the pipe allows the device to be securely positioned in the center of the pipe avoiding contact of the TC and BI with the surfaces and/or condensates.

It is known that folded glassine has the potential to affect the D-value of the BI. The reproducible “S” rolling shape of the strip into the MeCo Round has been designed to avoid folded glassine layers; and allows steam to come into contact with the entire surface of the strip. Any risk of post-process contamination is also avoided as the BI stays protected in the MeCo body from the recovery until it is transferred to the microbiological laboratory and opened under aseptic conditions to be cultured.
Damage of the process equipment and material remnants will be prevented, since all MeCo solutions are produced of soft materials (which are certified Pharma approved USP Class VI) that do not leave residues. When placed into the MeCo body the Mesa spore strip will be protected from shredding and it will never make direct contact with the surfaces of the process equipment. This will prevent ink remnants from being left behind in the process system.
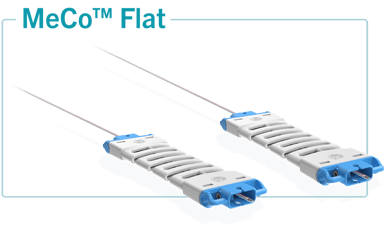
The MeCo Flat tool provides a solution for the placement of BIs and TCs during the validation of tanks and other equipment such as fermenters, freezedryers, and autoclaves. MeCo Flat ensures safety with secure placement of the BI and the TC without the use of tape or other means that could compromise the entire installation’s integrity, while ensuring reproducibility of physical and biological measurements. The adhesive used on the MeCo Strip, does not leave any visible residue or small particles behind in the system.
MeCo is a single use, recyclable device, to ensure security and to comply with high quality standards. The placement of both the BI and the TC is perfectly controlled and reproducible. The integrity of the piping system is preserved by limiting the contact of external items. Temperature measurements are controlled without the risk of erroneous measurements caused by surface or condensate contact. In the same way, the BI will maintain its integrity throughout the entire cycle eliminating contact with the condensates, the use of tape or other elements that may prevent steam-spore contact, and the risk of BI loss.
MeCo can greatly reduce erroneous results, eliminate unnecessary repeat cycles, and provide confidence in your validation process. SIP validation has never been so easy!
About the Authors
Written by Silvia RIZZI and Laurent BERLIET, from MESALABS France- Sterilization and Disinfection Control Division.
Laurent began working in 2010 as Technical Sales Rep with Amilabo which became Mesa Labs France. He holds a Biology and Geology License from university of Lyon, France. Silvia began her career with Mesa Labs France in 2016 as a Technical Sales Rep. She holds a Chemical Eng. MSc from University of Naples, Italy.
They support and train companies within the Pharmaceutical, Biotechnical and Medical Device Industries finding consistent solutions to validate sterilization and decontamination processes.
